Bernoulli-exponential survival model for estimating proportions in randomised controlled trials with dropouts
Keywords:
Bernoulli distribution, Exponential distribution, Clinical trials, Survival models, CensoringAbstract
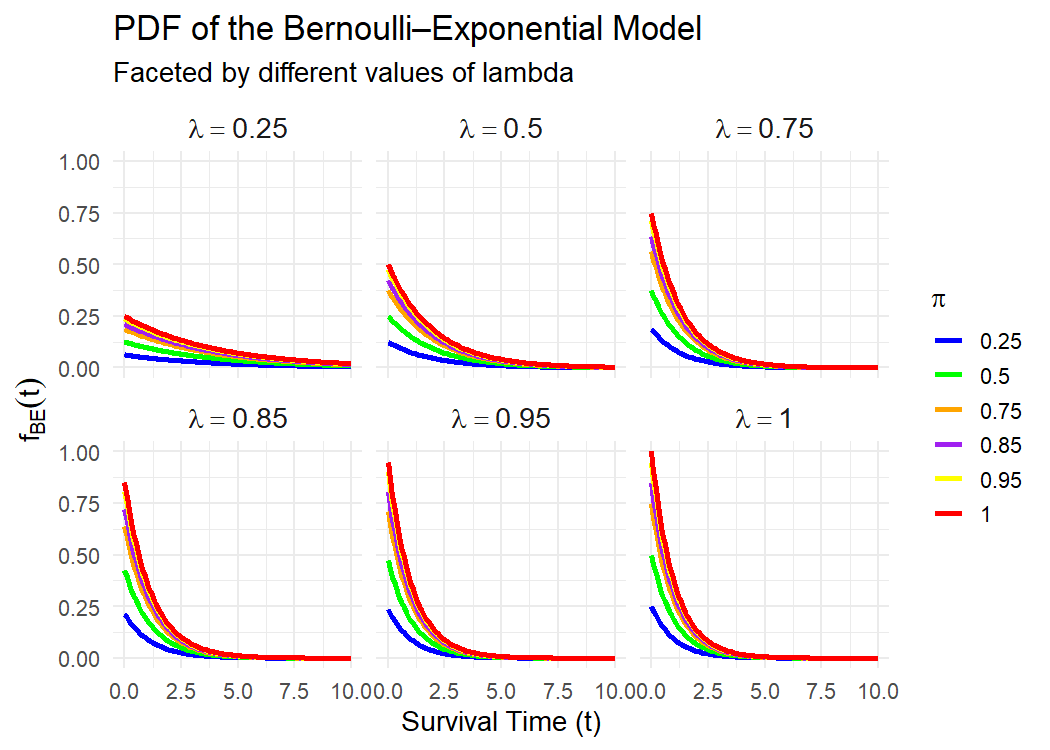
Randomised controlled trials often experience dropouts, especially in multi-stage designs where participants must meet induction-phase eligibility requirements before progressing to the main study. Loss of information arising from early non-responders or withdrawal of clinically vulnerable subjects are not always random. Classical survival analysis methods including the Kaplan-Meier estimator, exponential models, and similar techniques assume independent censoring. When this assumption is violated, survival probabilities are overstated, hazard rates are underestimated, and median survival time become biased. Such dropout mechanisms also lead to biased estimates of the proportion of participants who would experience the clinical endpoint, thereby distorting treatment effect measures based on event counts. This study proposed a Bernoulli-Exponential (BE) model that jointly represents the occurrence of an event and the time to event, rather than treating censored subjects as if they share the same risk structure as event completers. The model incorporates a Bernoulli component to describe the probability of belonging to the event-generating subgroup and an exponential component to model event times conditional on experiencing the event. This separation enables the model to account for informative dropouts, induction-phase failures, and scenarios where some participants contribute no event time. We derive the parameter estimates, survival function, and outline procedures for estimating the median survival time, its variance, and confidence interval. Simulation studies were used to assess bias and precision under varying levels of dropout. The BE estimator consistently corrected the upward bias observed in Kaplan-Meier, intention-to-treat, and completers-only estimates under heavy or informative dropout.
Published
How to Cite
Issue
Section
Copyright (c) 2026 Ekele Alih

This work is licensed under a Creative Commons Attribution 4.0 International License.







